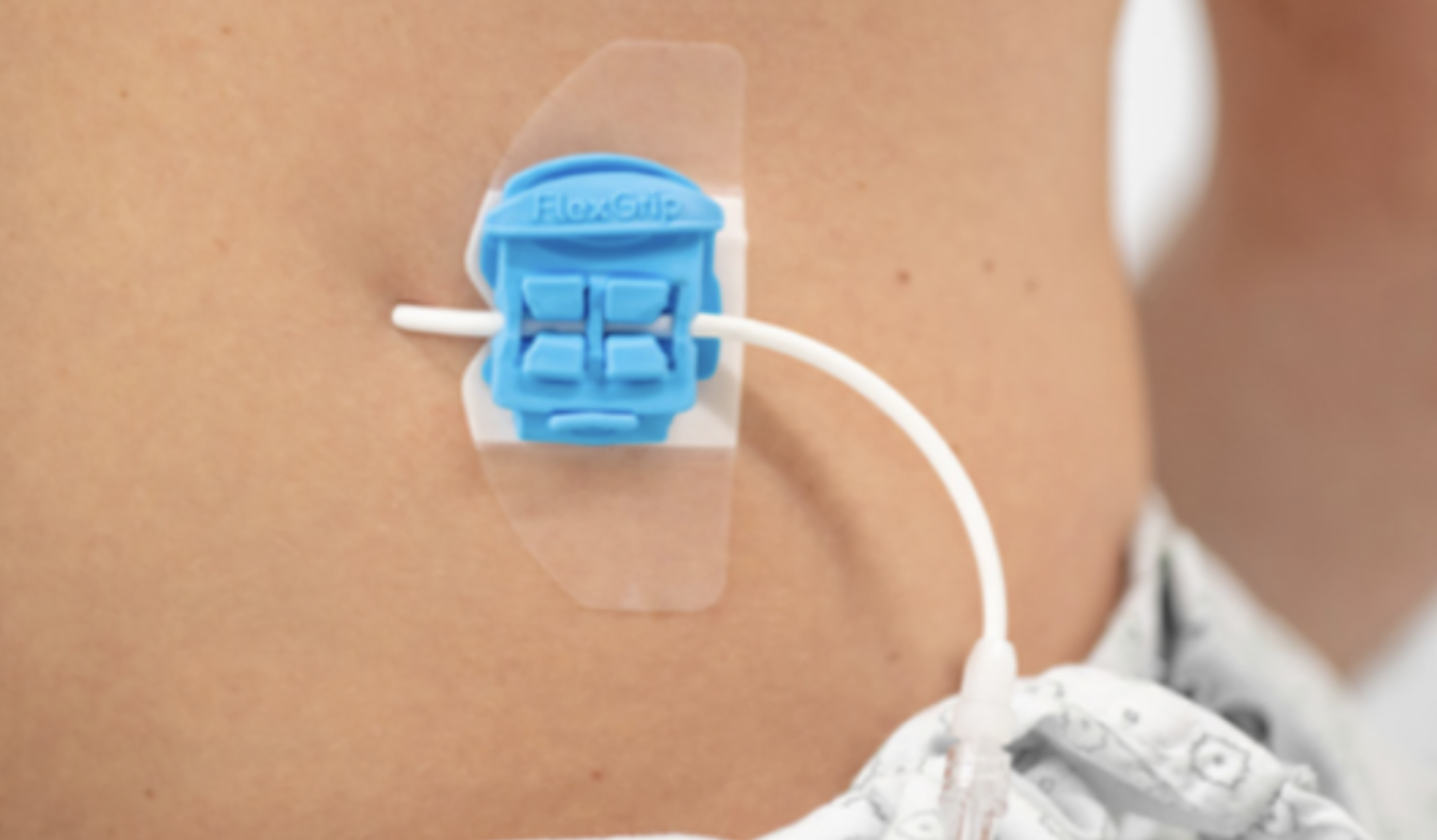
FlexGRIP Universal 5-11 Fr
FlexGRIP®
FlexGRIP Universal 5-11 Fr suitable for stabilizing & fixating a large range of catheters, such as: nephrostomy (post-op), drainage catheters, gripper needles (Port-a-cath), thrombolysis catheters, jejunostomy, tubings, cables and drivelines (eg. LVAD).
Features
-
large diameter range: 5-11 Fr
-
unmatched strength of securement
-
easy to use; pre-mounted fixation leash
-
additional lock for leash
-
skin-safe hypoallergenic adhesive
- soft materials only
Benefits
-
universal system; wide variety of applications
-
designed to:
-
prevent catheter migration & related complications
-
prevent MARSI – medical adhesive related skin injury
-
optimize patient comfort
-
-
extra safety lip prevents accidental loosening
-
extra patient comfort
Intended use: The device is used in combination with a compatible catheter, tubing or cable. The device consists of an adhesive dressing and a clamp part to reduce catheter migration and pull out. In combination with a catheter or tubing it is to be used specifically for therapeutic purpose and intended to be used for treatment or alleviation of disease. The bedal catheter stabilization device is a single use device and is attached to the intact skin for a maximum of 7 days.
Intended Users: The device is intended to be used by trained medical professionals in a hospital or clinical setting.
Contraindications: Known hypersensitivity or allergy to hydrocolloid materials or any component of the device.
Safety considerations: Discard if package is damaged.
Key points to the Bedal catheter stabilization device usage:
-
Prepare the skin according to the standard hospital protocol for dressing application. Allow the skin to dry completely. Skin prep or hair removal may be required on some patients for better dressing adherence.
-
Remove any residue of the dressing pad with alcohol after removing the device.
-
The extra strip can be used to temporarily secure the catheter during dressing
change.
- Discard after use in appropriate containers for potentially infectious material.
Warnings and precautions:
-
Do not use the device where loss of adherence could occur, such as non-adherent skin or confused patient.
-
Always follow the infection control guidelines at your facility.
-
Minimize catheter or cable manipulation during application and removal of the
device.
-
The device and the surrounding area should be routinely inspected.
-
Use sterile technique for application and removal of the device.
-
In case of loss or di_erence in performance, such as loosening of adherence to the
skin, the device should be removed and replaced.
- Read all package insert warnings, precautions and instructions prior to use. Failure
to do so may result in severe patient injury.
Contact Information:
Nexaso Medical Co., Ltd.
47/316 Kaitak Tower, K005010, 5th Floor, Popular Rd, Ban Mai, Pak Kret, Nonthaburi 11120
Tel. 02-0175-766
License No. ฆพ. 357/2569